So there is a Mr. A?
Preseason football brought methicillin-resistant staphylococcus aureus (MRSA) back into the limelight with multiple Buccaneers coming down with serious infections. Thanks to Twitter’s unfiltered access to everyone, this news came out via an airing of grievances over missed diagnoses and the team’s subsequent glossing over of Lawrence Tynes’ recovery (interesting aside, he’s filing a grievance over being placed on the non-football injury list, turns out it might not be the best idea to publicly blast the team but at least we get that wonderful header pic). Less recent examples that leap to mind are the string of infections that plagued the Cleveland Browns for half a decade. Professional sports aside, MRSA and its encompassing family of staphylococcus aureus bacteria are a monumental health concern, especially as a source of illness from within the healthcare system.
According to the CDC, data from 2005 shows “an estimated 478,000 hospitalizations with a diagnosis of s.aureus…of those approximately 278,000 hospitalizations were related to MRSA.” This next bit is a little worrisome but I promise it’s not as bad as it sounds. In 2003, 29% of people were colonized with s.aureus and 1.5% with MRSA. Many of you reading this are doing so with s. aureus bacteria happily filling your nose. Don’t panic. It’s often asymptomatic, bacteria is everywhere (literally coating everything), and lots of it is s. aureus. I’ll do my best here to explain what s. aureus and MRSA actually are, why they make us sick, how our bodies defend themselves, and why these bugs are so problematically stubborn. I’d also like to apologize, that’s a lot of very dense ground to cover. I’ll do my best to keep this interesting but micro can get pretty dark.
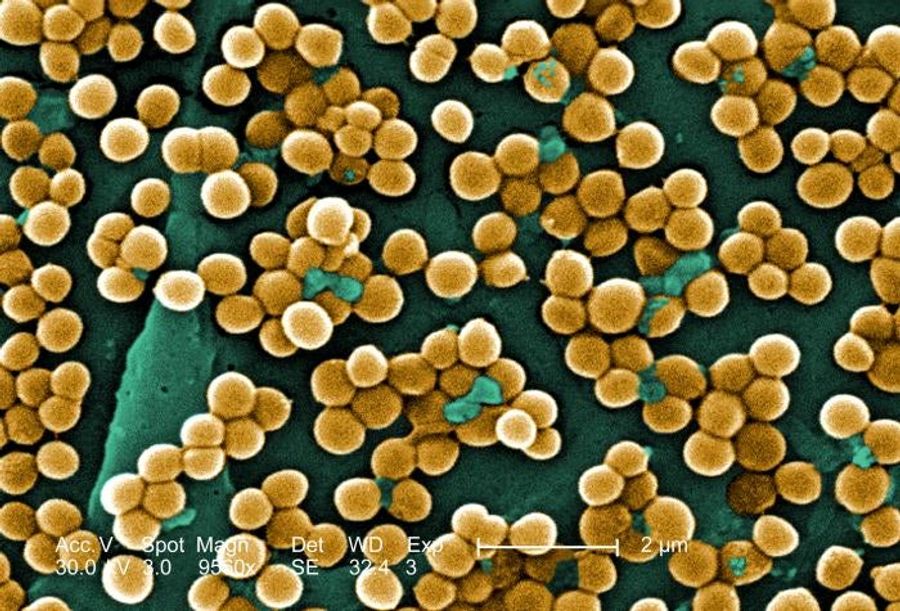
Anytime you hear about an infection or bacteria with “cocci” or “coccus” in the name, picture something like you see above. That term refers to spherical bacterium, although the clustering shown above is unique to staphylococcus (staphyle is Greek for “cluster of grapes”). Like I said before, it might be living on you now, a sheet of bacteria with nothing but your skin and immune system standing in its way. Luckily, your skin is really quite good at keeping the outside outside. We’ll assume the worse though, that s. aureus gets past your skin, and start looking at some of the special characteristics contributing to both the bacteria’s continued survival and your growing symptoms.
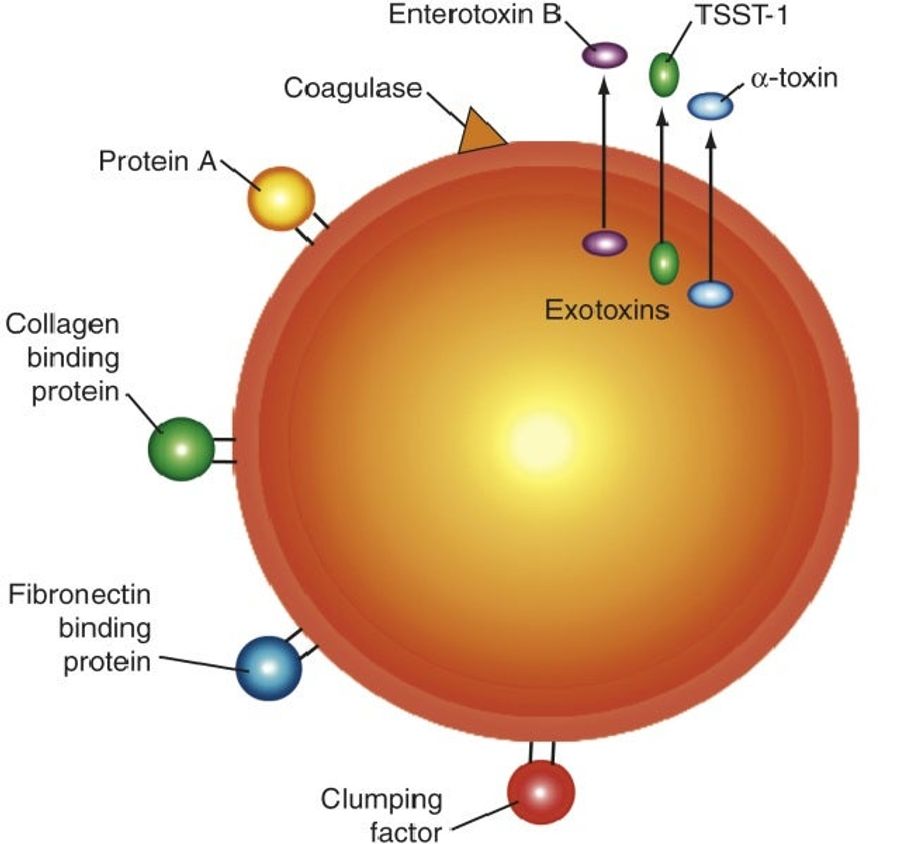
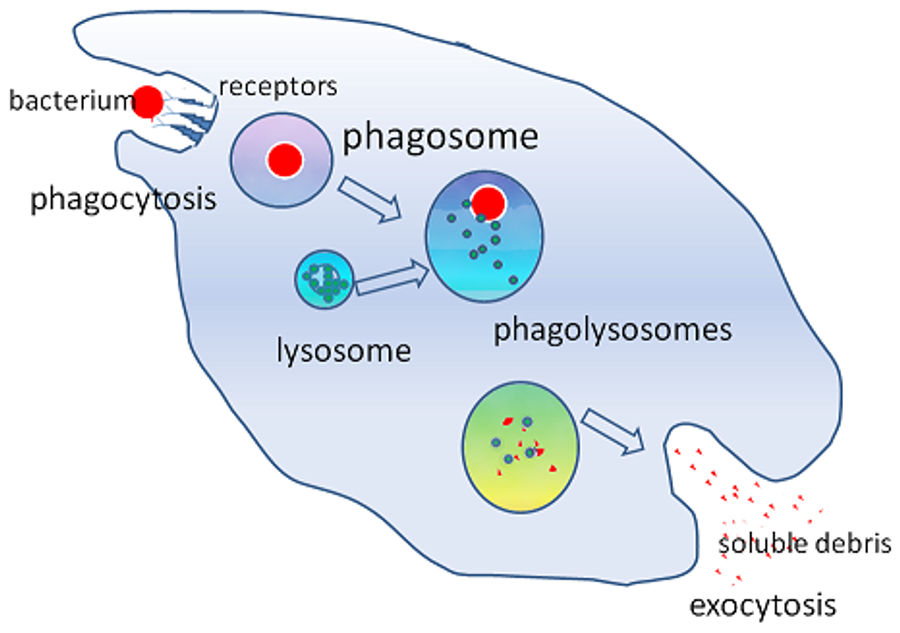
This is gratuitously oversimplified, but that’s what we’re going for here. Each of the specific molecules on the diagram above contribute to the virulence (or pathogenecitity/survivability) of staph infections. First, this bug has to protect itself from your immune system. As soon as it’s past the skin, your body’s ubiquitous immune molecules will go to work. One of your major defenses is opsonization. Simply put, a collection of immune system molecules will bind to exterior proteins of a foreign molecule. Their enveloping presence triggers macrophages (think garbage trucks of the immune system) to phagocytize (think eat) the bacteria. This process is pictured below, probably best if you ignore the words and just pay attention to what happens to the red bacterium.
Now back to that diagram with all the molecules on the bacterial cell wall. Protein A extends from the surface of bacteria and will bind the enveloping opsonization molecules (IgG antibodies) in a way that inhibits their pro-phagocytic signalling. In a sense, they use the body’s own signalling molecules as camoflage to hide from the immune system’s search dogs.
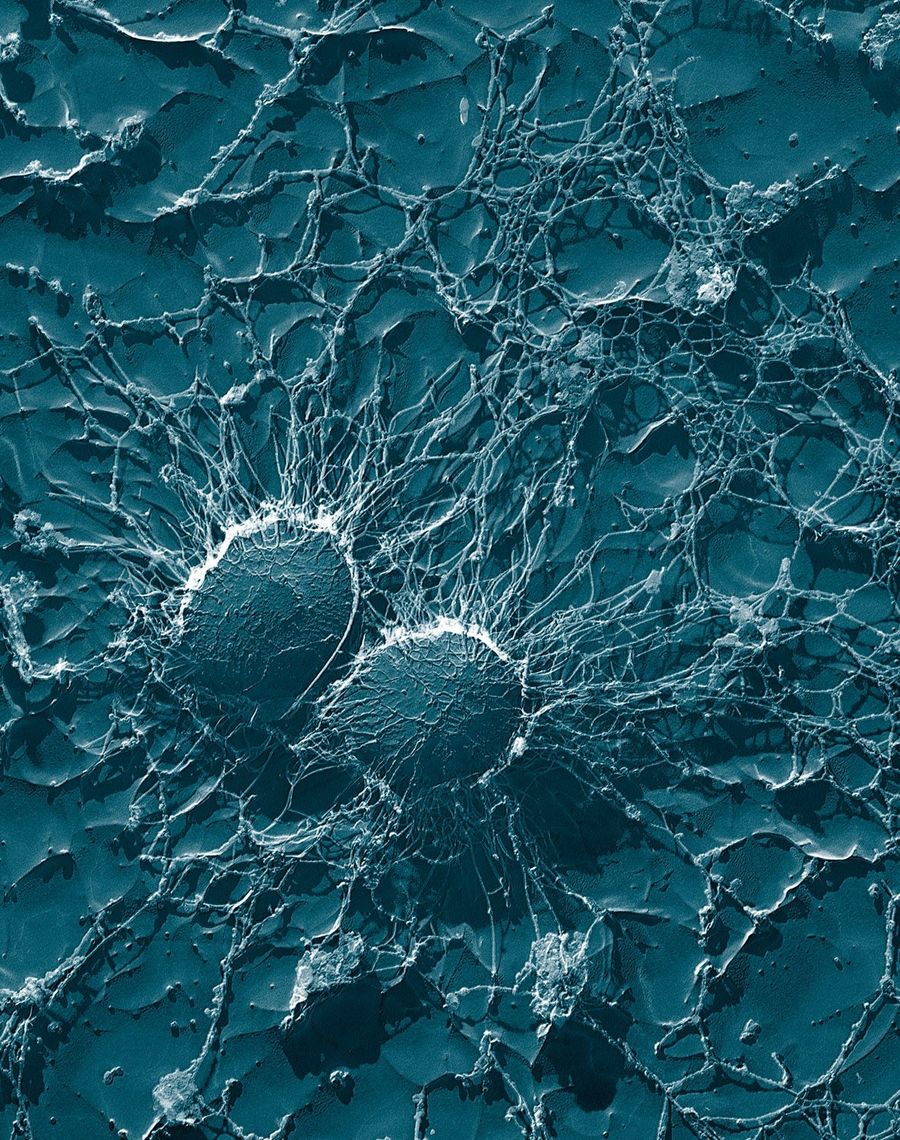
Redundancies are lifesavers in every body system, and the immune system is no exception. The diversity in signaling molecules and cellular defenses have driven bacteria to adopt equally impressive countermeasures, including coagulase. Coagulase triggers one of the final steps in our regular clotting cascade (a series of enzymatic reactions that ends with your blood clotting in wounds), and along with clumping factor, surrounds the bacterium with fibrin, a component of blood clots. The picture below is an electron microscope scan of s. aureas surrounded by a protective fibrin meshwork in cow blood. This meshwork serves as a physical barrier inhibiting the binding of your body’s various immune cells.
There are other features helping protect staph from your intrinsic immune defenses, but we’ll move on to how it actually makes you sick. Staph can be expressed as skin infections, endocarditis (infections commonly on the heart valves), osteomyelitis (bone infections), pneumonia, or systemically in bacteremia and sepsis. In systemic cases, infected abscesses can form in any organ, but now we’re just piling on. The horrible illnesses attributed to staph infections largely arise from exotoxins the bacterial colonies will produce and release. It’s worth noting these protective measures and toxins aren’t all expressed by all staph bacteria, which also explains the incredible diversity of staph infection symptoms/locations.
Toxins α, β, γ, and δ are all hemolytic toxins, meaning they disrupt the membranes of blood cells, destroying them in the process. α toxin does so by forming an open pore in the blood cell membrane. Passage of water and molecules through cell membranes is incredibly fine-tuned and this deregulated traffic kills the cells.
The remaining toxins deserve their own special designation as superantigens. We’ll think of antigens as any molecule or protein with which your immune system’s antibodies can bind. This binding is often specific, which is why your body can develop unique antibodies to specific illnesses (why you don’t get chicken pox or mono multiple times, why vaccines work safely…WHY VACCINES WORK SAFELY). However, superantigens are capable of non-discriminate activation of your immune system’s T-cells. This activation triggers massive T-cell regulated release of cytokines, molecules that mediate immune and inflammatory responses. Simply put, this is how a bacterial infection causes systemic shock: plummeting blood pressure, high fevers, elevated heart rate, hyperventilation, organ failure, and death. This can be trouble. You need to be in a hospital getting fluids and IV antibiotics right away.
Enterotoxin A is the primary cause of food poisoning and will run your intestines through a gamut of diarrhea, vomiting, and cramping. While it is a superantigen, these reactions will usually be self-limiting to the intestines and GI tract. You’ll be in a rough way for a while and could need some IV fluid management, but the illness should run its course without too much trouble.
Toxin Shock Syndrome Toxin 1 (TSST-1) surprisingly causes toxic shock syndrome. This syndrome will typically present as some collection of a sudden high fever, low blood pressure, confusion, seizures, headaches, etc. Strangely enough, nine times out of ten the patient will be a woman either menstruating or having just finished menstruation and using tampons. If you’ve never heard of the defective tampon fiasco of the late 1970s, allow me to enlighten you. Nowadays this is extremely rare, better tampons I guess.
Finally, we have the exfoliative toxins. These toxins cause Scalded Skin Syndrome, which presents mainly in infants and children under 5 and causes the outer layer of skin to slough off. Large fluid-filled blisters form and the patient has a positive Nikolsky’s sign, which is a very cold, clinical way of saying their skin falls off if lightly rubbed. Thankfully, prognosis is good and assuming treatment of the original infection and dermatological symptoms, the child will fully recover with minimal scarring. Take a minute here to step back and realize just how insane this is. There’s a small unicellular organism that is phenomenally successful from an evolutionary viewpoint. It’s fucking everywhere. 30% of human beings on earth have millions of them living in our noses (that’s near 2 billion people). If you’re immunocompromised because of chemotherapy, or you fell off a bike and had your spleen removed, or you have AIDS, or you’re not immunocompromised but just impressively unlucky, this infection grows inside you. Now flourishing inside you, it produces a waste molecule. Staph doesn’t use this molecule for food, or to guarantee its survival, it just makes it, a by-product. However, this particular toxin cleaves one particular protein (desmoglein-1) that happens to hold two layers of our skin together. End result: outer layers of skin fall with the slightest provocation (like gravity or an infant moving because they don’t understand not to move).
Good news, that’s it for suffering infants or food poisoning or tampon-induced systemic shock and the following history of how we ended up with resistant strains should be lighter reading. Staph was first identified in the 1880s, a long time ago. The following decades saw increasing use of antiseptic protocols, but understanding of the vectors transmitting infection and disease was limited and treating staph (or any infection) was a problematic endeavor. Then penicillin was discovered around 1930 and we cured all infections immediately without any unforeseen side effects ever. The medicinal use of penicillin was gradually worked out with mass production expedited for World War II.
You’re likely familiar with the problems here. Bacteria reproduce quickly. Assuming a receptive growth media and proper nutritional factors, staphylococcus aureus populations commonly double in less than an hour. This rapid growth allows for successive generations to quickly produce genetic diversity and increases the frequency of mutations. Throwing penicillins at this quickly adapting, resilient population killed large swaths of bacteria, but left ample growing conditions behind for those lucky strains conferred resistance. To make things worse, resistance-providing genes could be encoded in transferable plasmids (small circular extra-chromosomal bacterial DNA) or picked up by bacteria-infecting viruses (prophages), both mechanisms of genomic transfer from one bacterial strain to another.
Before long, strains of staphylococcus aureus were showing up with penicillin resistance. A stronger version of the antibiotic, methicillin, was produced in 1959 for treatment of these persistent bugs. This time it only took two years for s. aureus to develop resistance. I promise I’ll avoid chemistry generally but explaining the significance of methicillin resistance requires just a quick look at penicillin’s structure.
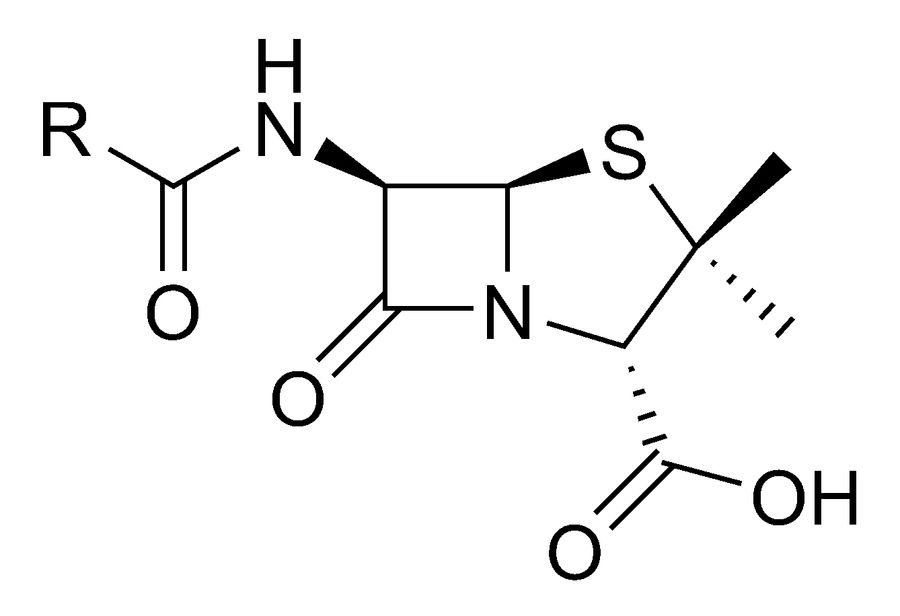
See those two rings? There’s a five-member ring connected to a four-member ring. It’s corny, but think of them as a house (5-member) and a garage (4-member…the square with an N in the corner for the non-chemistry folk). The garage is called a β-lactam ring, and it’s important. That ring provides penicillin’s antibiotic nature by binding to structural components of the bacterial cell wall, breaking them up and killing the bacterial cells. There’s a whole list of penicillin-like drugs called β-lactam antibiotics: methicillin, penicillin, oxacillin, dicloxacillin, and more. MRSA carries a gene, mecA, which changes the target receptor of the antibiotics into a form with significantly less binding affinity, causing these β-lactam drugs to all effectively ignore MRSA.
We’ve learned from our mistakes and antibiotics are now used with less reckless abandon. If your doctor says, “I think it’s a virus, rest and fluids for a few days and if you’re still sick in three days come back to see me,” there’s probably a good reason. When possible, even MRSA infections are treated without antibiotics. Many skin infections can be treated with proper wound care, but more severe or widespread infections do require more aggressive treatment approaches. Sensitivity tests are run on bacterial cultures and antibiotic treatments are tailored for the specific infection at hand. These precautions help prevent unnecessary use of antibiotics and preserve their effectiveness.
Hospitals still pose a problem. Considering the strain’s stubborn resilience, it’s hard to think of a better home than a hospital full of catheter and IV lines providing access to immunocompromised patients, often elderly, staffed by a team of medical professionals who can serve as healthy, asymptomatic carriers. This environment, conducive to growth and especially dangerous for already ill patients, has resulted in the proper implementation of infection protocols in healthcare settings being vital. Rapid identification, isolation, and treatment of MRSA infections is hugely important for minimizing the impact of these infections, and making sure hospitals remain a place you feel comfortable visiting to get healthy.
This improved approach has been working, and hospital-acquired (or “nosocomial”) infections have been decreasing. More importantly, severe systemic blood-stream infections have decreased at a greater rate. Unfortunately this doesn’t have a clear cut resolution somewhere in the future, the stronger drugs we create, the stronger bacteria will become to survive. This balance will always be a major influence when creating widespread treatment protocols. Hopefully the lessons we’ve learned (don’t play for the Browns) and appropriate use of newly developed drugs will help us make continued strides controlling and containing the most deadly of these infections.
- Best NBA Betting Picks and Predictions for Monday April 6th
- National Championship Bet Pick: Why Michigan Has the Edge Over UConn
- UFC Vegas 115 Betting Picks: Moicano vs. Duncan Headlines April 4th Card
- NBA Betting Picks April 4th: Three Best Bets for Saturday's Slate
- Michigan vs. Arizona Bets: Wolverines Hold Edge in Final Four Showdown
- Best NBA Betting Picks Today: Friday April 3rd Expert Predictions
- MLB Pitcher Props Today: Best Baseball Bets for April 3rd












